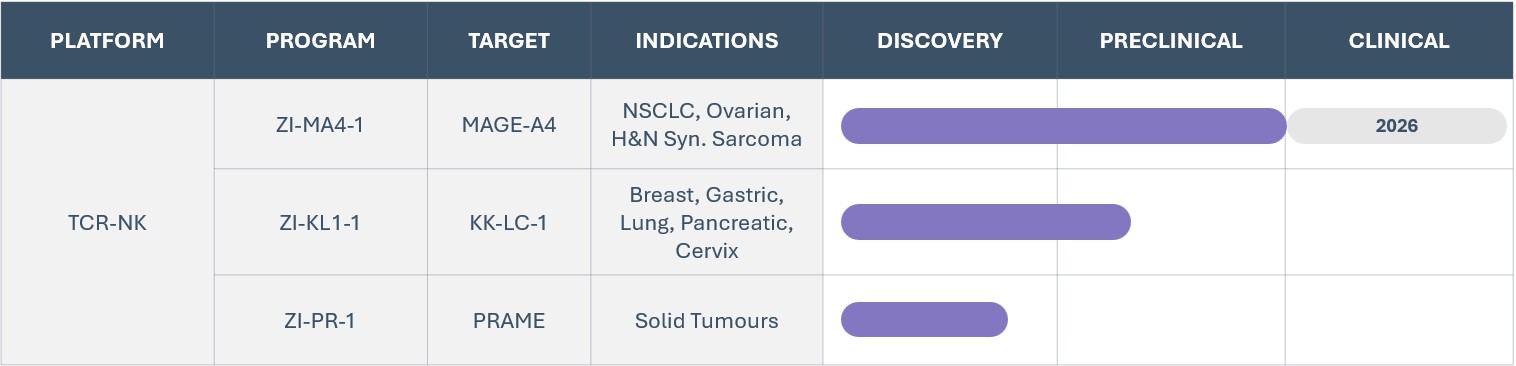
Zelluna’s pipeline assets target a blend of antigens that are either clinically or preclinically validated and expressed across a broad range of solid tumor indications, providing high potential for patient impact and a huge market opportunity.
- MAGE-A4 and PRAME are clinically proven TCR targets for solid cancers; one market approval for MAGE-A4 targeting agent and PRAME targeting agent in registration study.
- KKLC-1 is a preclinically validated solid cancer target.
Positive regulatory interactions as well as plug-in manufacturing process apply to the entire pipeline and platform de-risking concept and development path for all pipeline programs.